CYSTADROPS is the first and only viscous ophthalmic solution with practical, 4 times a day dosing for the depletion of corneal cystine crystals1
CYSTADROPS contains 0.37% of cysteamine (equivalent to 0.55% cysteamine hydrochloride). CYSTADROPS is formulated with carmellose sodium for greater viscosity.1
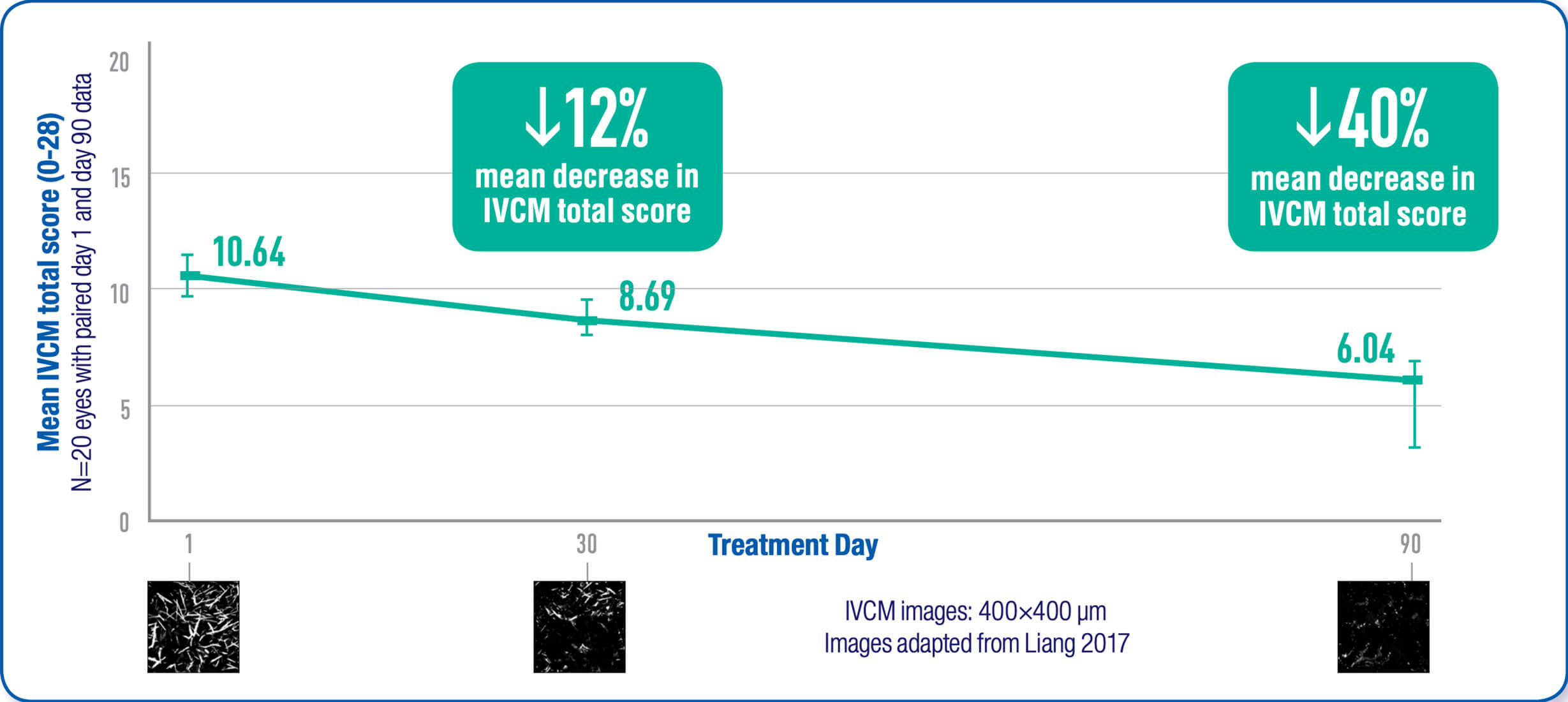
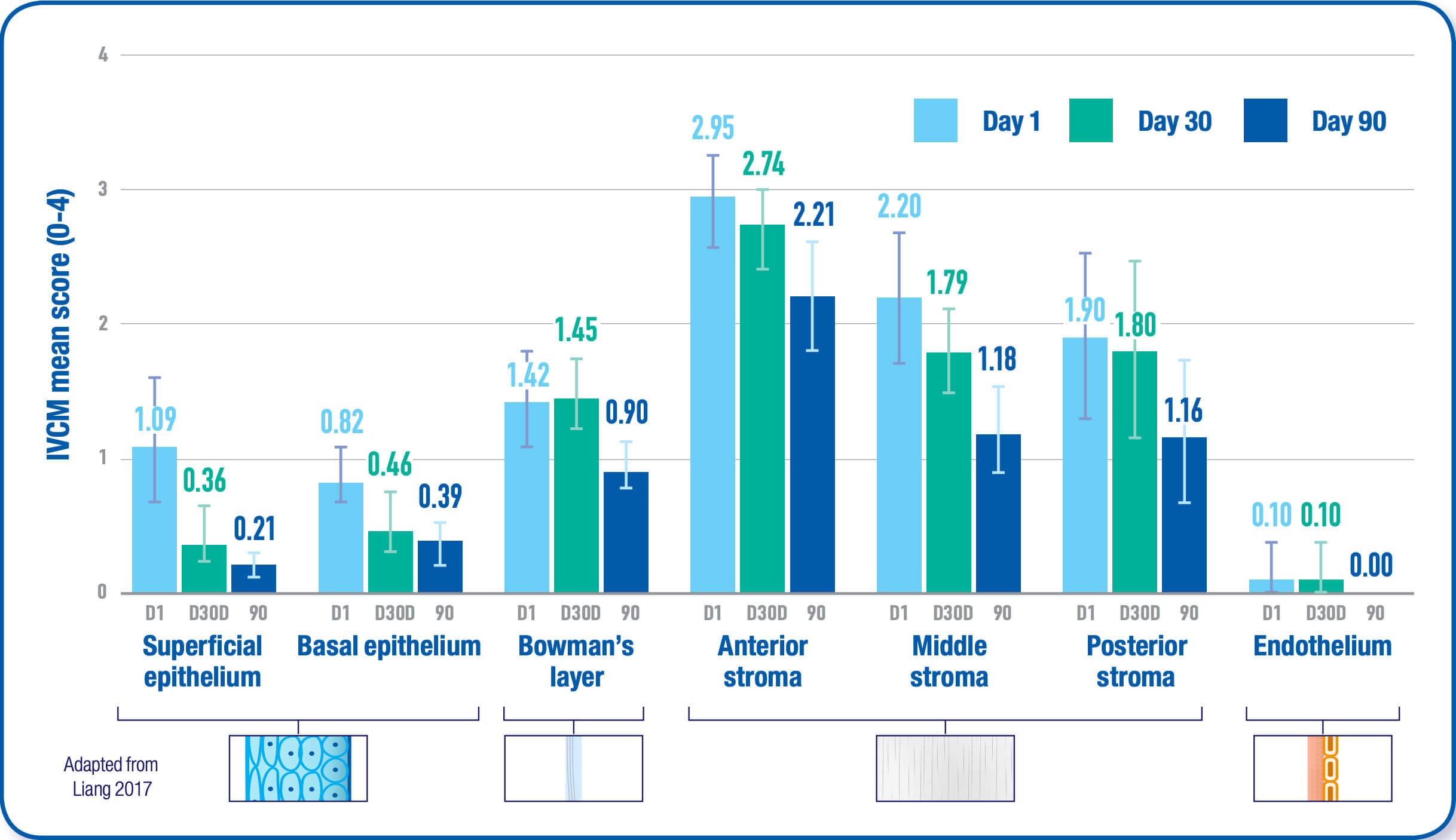
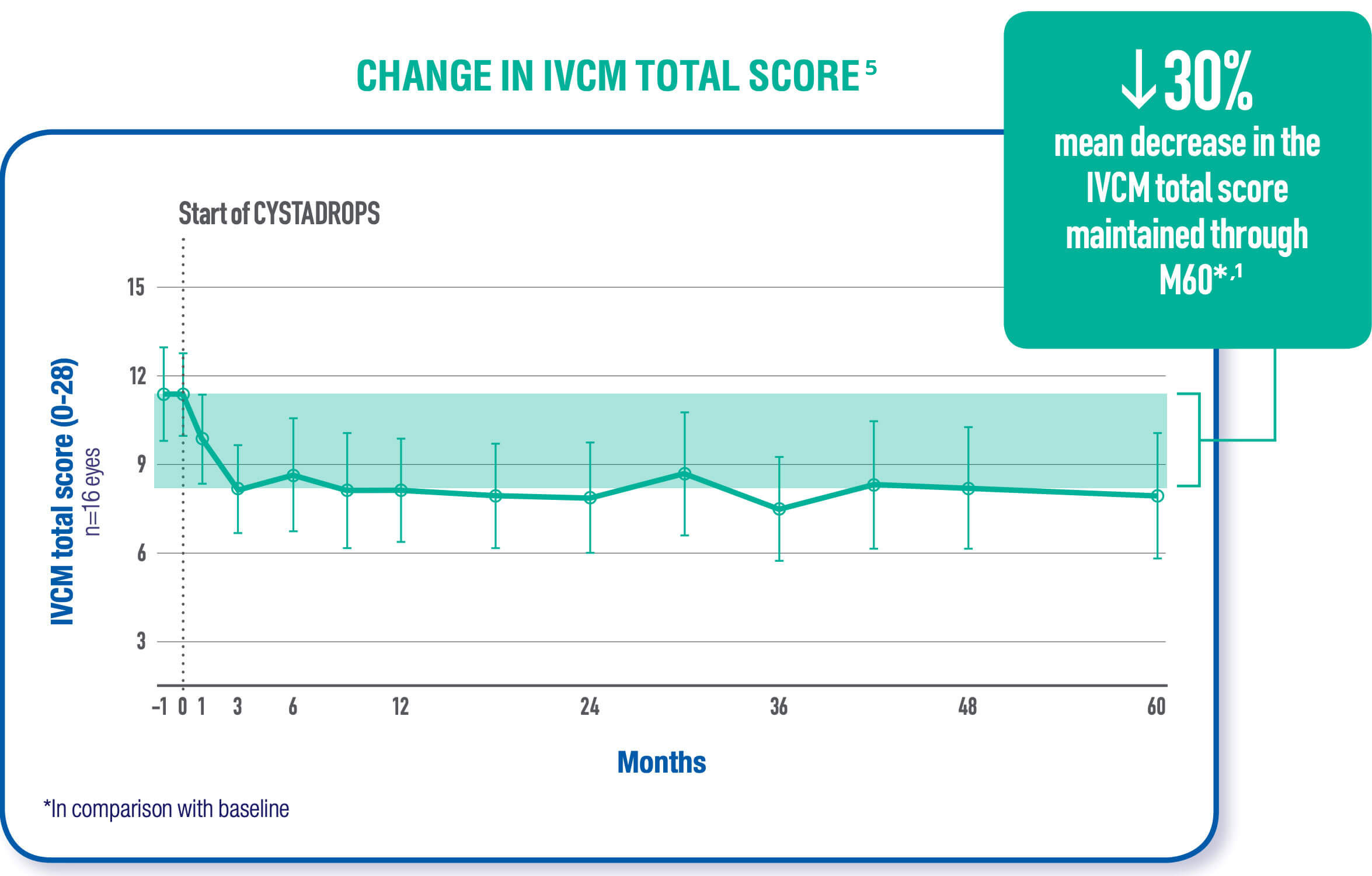
Viscous cysteamine coats the cornea allowing the active ingredient to penetrate the cornea cells. The cysteamine converts cystine to cysteine and cysteine-cysteamine mixed disulfides which can then exit the lysosome. As a result, corneal cystine crystal accumulation is reduced in all layers of the cornea with continued use.1,2
Cystadrops contains benzalonium chloride, which may be absorbed by soft contact lenses. Contact lenses should be removed prior to application of solution and may be reinserted 15 minutes following its administration.1